Part.2 : SULFATES
SULFATES
Sulfur is somewhat unusual in that it serves as anion in sulfide minerals & cation in sulfates
In sulfates, sulfur atoms lose all 6 of outer e-
4 from 3p & 2 from 3s subshells
These small high-charged cations coordinate with 4O anions to form SO4-2 groups
Sulfur-oxygen bond stronger than oxygen & other cations bond, anisodesmic

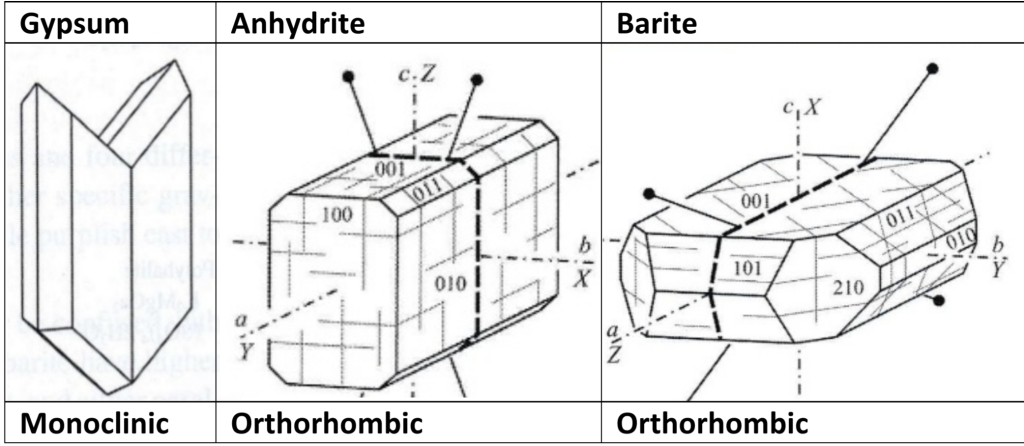
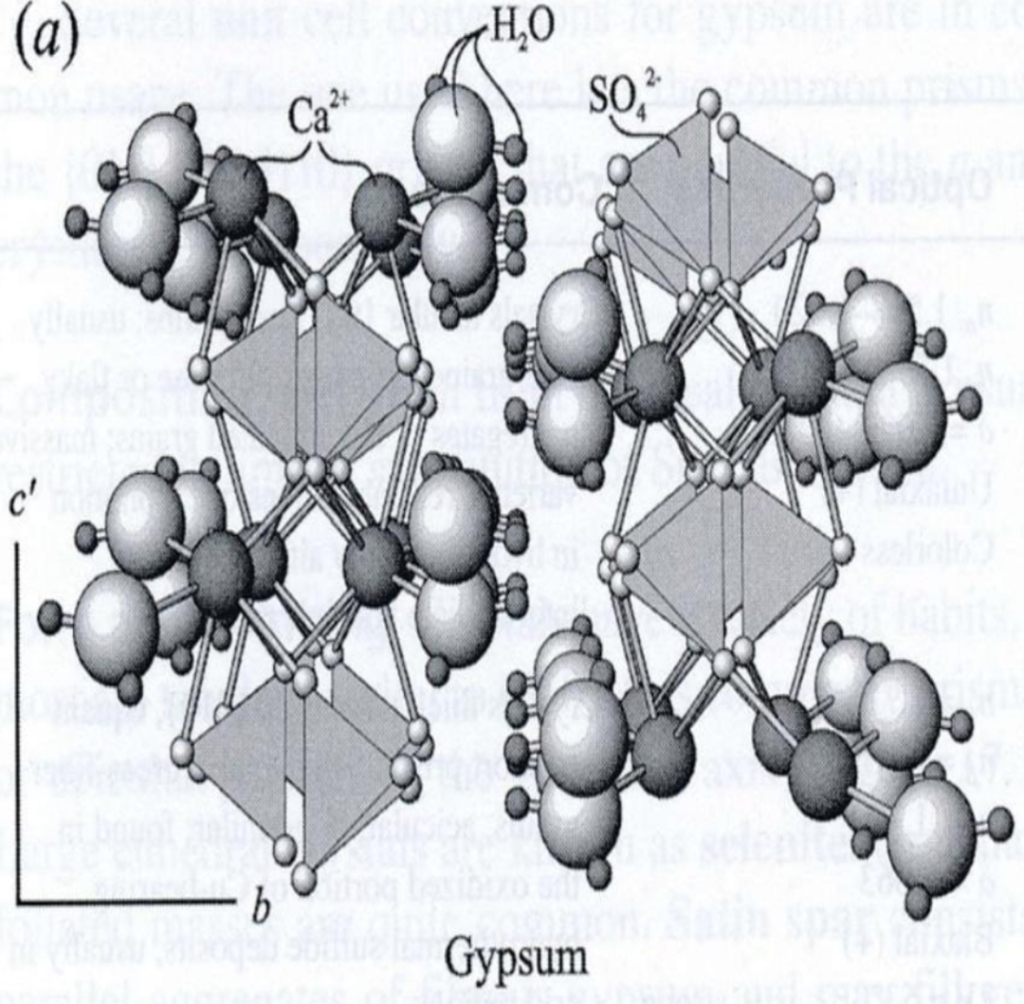
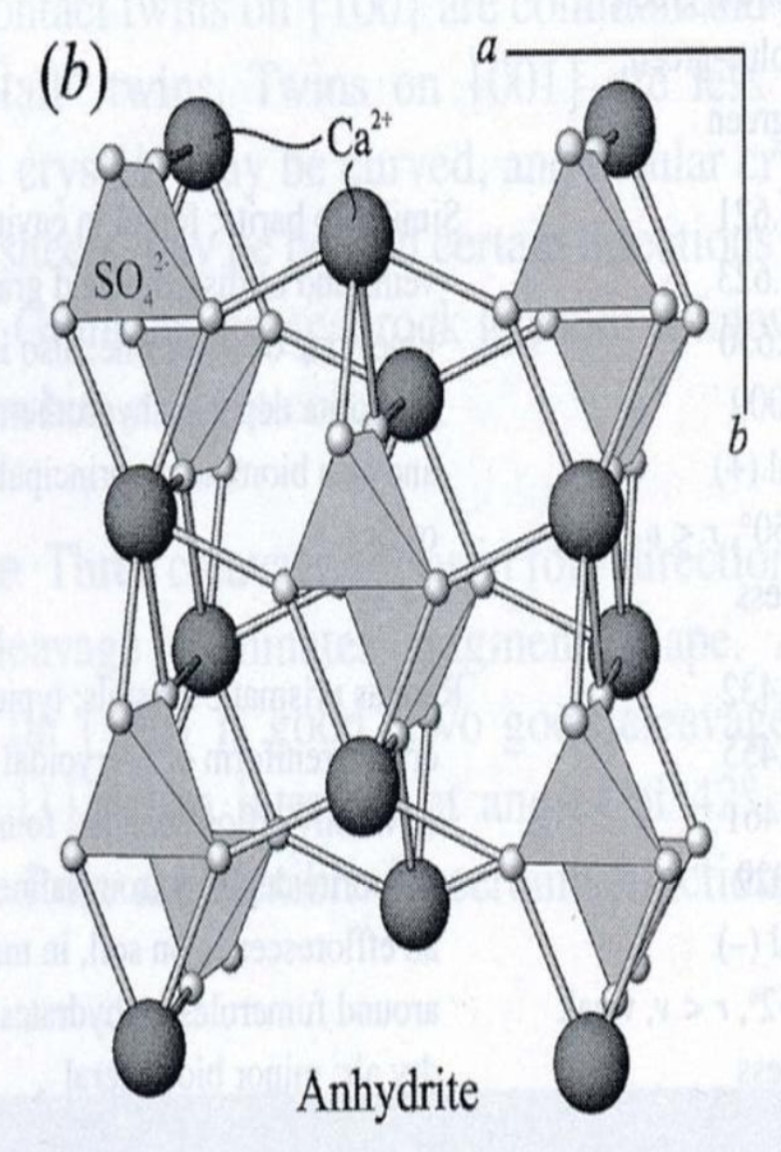
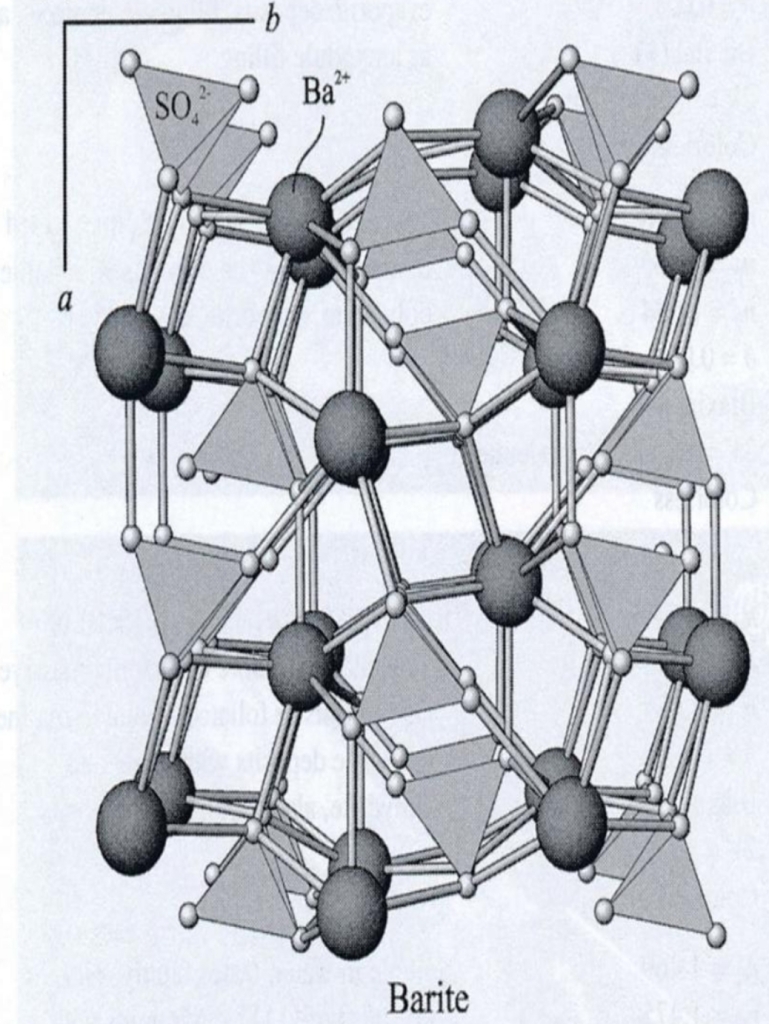
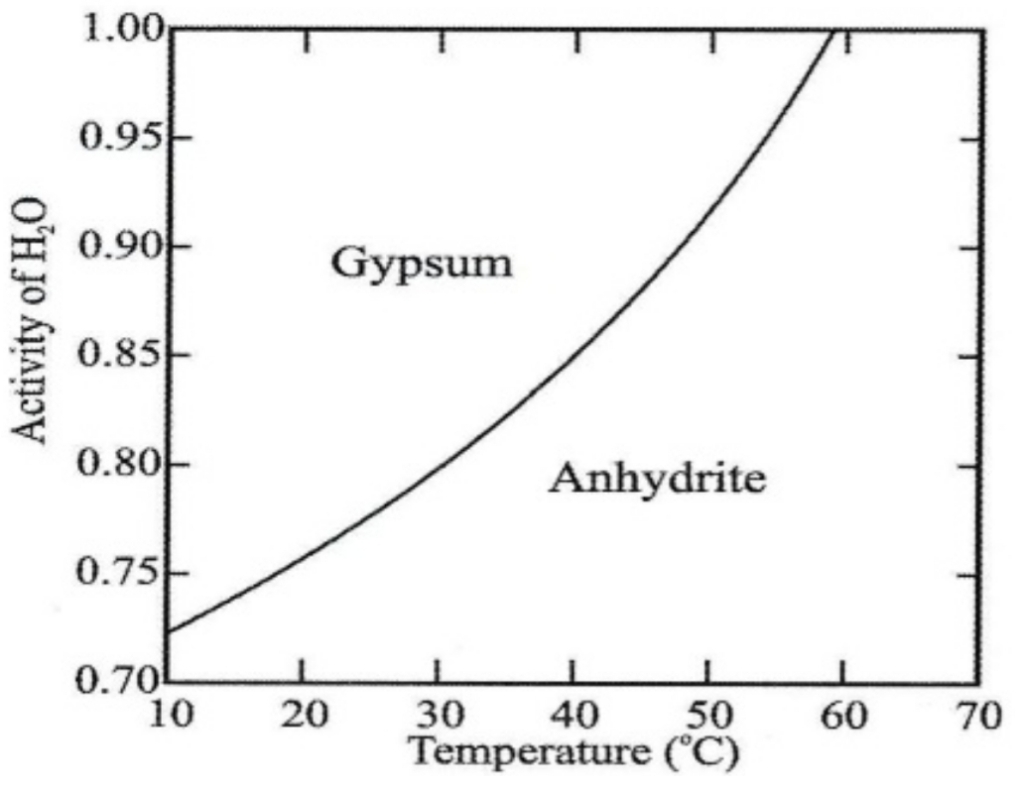
Composition usually restricted to limited substitution of Sr or Ba for Ca
Gypsum is stable phase in evaporate environments, & under high salinity conditions or T, anhydrite is stable phase
